|
| Illustration: Leigh Nelson |
Scientists try to untangle the mystery behind a rare neurodegenerative disease. Natalie Jacewicz follows their twisting path. Illustrated by Leigh Nelson.
Tom Looney was speaking to top government security officials in 2007 when he began to feel dizzy. Looney, the industry director of homeland security and public safety for Microsoft, carefully descended the steps from the stage in Redmond, Washington. He stumbled back to the green room and passed out.
Assuming Looney had suffered a heart attack, his coworkers called an ambulance. But doctors at the hospital were puzzled. “You didn’t have a heart attack,” Looney recalls them saying, “but something’s not right.”
That was the last time Looney went to work, and the first of many days spent in hospital waiting rooms. He was 48.
Looney had developed a rare brain disease—an ailment that increasingly encroached on his day-to-day routine. As he wrestled with the disease in ensuing years, the ailment began to transfix the scientific community as well. Partly by accident, researchers in San Francisco think they discovered the cause underlying Looney’s condition: tangled meshes of protein. What’s more, they now believe similar tangles underlie Alzheimer disease, Parkinson disease, and other debilitating illnesses that wage war on the mind.
Scratching sheep
The recent research into Looney’s condition and others like it began in the 1980s with Stanley Prusiner, a neurologist at UC San Francisco. He was studying neurodegenerative diseases—ailments that attack the nervous system.
He focused on scrapie, a disease that causes sheep to stagger around pastures and scrape themselves against fences, as though scratching an insatiable itch. The same illness causes mad cow disease in cattle and Creutzfeldt-Jakob disease in humans. The disease is contagious; humans can become infected by eating contaminated beef. Prusiner wanted to understand how this contagion worked.
After subjecting scrapie-infected brain tissue to a battery of chemical tests, Prusiner discovered a baffling commonplace cause of contagion: proteins. Consisting of long chains of amino acids and found throughout the body, proteins fold into three-dimensional shapes determined by how the charged amino acids attract and repel one another. The complex folds hide parts of the protein and leave other sites exposed, dictating how each specifically shaped protein interacts with other molecules.
The proteins Prusiner identified seemed to co-opt this system. Through unknown means, these proteins refolded into unnatural contortions and then proselytized others, converting one protein after another into jumbles that mobbed the brain. Cells, unable to digest these disordered masses, lost function and died.
Prusiner proposed the word “prion” to describe the infectious proteins. He chose “PrP,” for “prion protein,” to name the proteins vulnerable to misfolding. Prions, he claimed, caused scrapie. But he didn’t stop there. Protein clusters in patients’ brains also typified other neurodegenerative diseases, like Parkinson and Alzheimer. Prusiner suggested prions might also set off those diseases.
Much of the scientific community, however, balked at the suggestion that a rebel protein could replicate itself without the help of DNA or RNA, the genetic molecules of life. “Prions have extraordinary biological characteristics, not the least of which is their possible non-existence,” says neuropathologist Laura Manuelidis of Yale University, one of Prusiner’s steadiest critics.
Prusiner stopped talking with reporters after several critical stories, but he continued to argue for prions, knowing many scientific breakthroughs initially seem heretical. “The incredulity of my colleagues only strengthened my conviction,” he wrote in his book Madness and Memory.
Other experiments gradually strengthened Prusiner’s claim, and most scientists grew convinced of the connection between prions and a small collection of diseases like Creutzfeldt-Jakob. But others thought prions might be a symptom of an infectious virus, not a cause themselves. Despite continuing controversy, Prusiner won the 1997 Nobel Prize in Physiology or Medicine. The committee usually honors teams working in broadly accepted disciplines. That year, Prusiner received the prize alone.
Labeling the disease
Ten years after Prusiner’s prize, Looney’s doctor visits began. A married father of two sons, Looney credits college basketball for enduing him with determination—which he later applied to his career at Apple and Microsoft. Now, he turned that determination toward discovering his disease.
When doctors asked him about his symptoms, Looney catalogued a sundry list of seemingly unrelated bodily rebellions. He could no longer crisply cap his words with consonants, and his slurred speech morphed into difficulty swallowing. His bladder felt constantly full, and when he stood up to his full height of 6 feet 5 inches, Looney sometimes fainted.
Finally, he visited a doctor who recognized the bizarre symptoms. Looney probably had multiple system atrophy, or MSA—a fatal disease causing nerve cells to lose function and die. When delivering the diagnosis, the doctor began to cry. So did Looney’s wife. Looney did not.
“I told myself, ‘You’re an athlete. You endured six years working for Steve Jobs,’” says Looney. “To me, it was a relief because now we could put a label on this thing.”
|
Natalie Jacewicz talks with Tom Looney, a long-term survivor with the rare neurodegenerative disease MSA, about his daily strategies to maintain his health. Click on image to play. |
While Parkinson disease strikes about 1.6 percent of the elderly population, MSA afflicts just 3 in every 100,000 people over age 50. But MSA kills patients twice as quickly. On average, they live no longer than 10 years past diagnosis. There is a variable mix of symptoms, including slurred speech, flailing limbs, and choking. No definitive test proves someone has MSA, so these symptoms offer the best clues to flummoxed doctors.
“This is a really challenging disorder,” says neurologist Kathleen Poston of Stanford University. “I’d say 90 percent of my patients are misdiagnosed the first time they’re seen by physicians. It can take years of seeing doctors before patients are diagnosed with MSA.”
Even with that verdict, doctors can do little to help. No known treatment cures MSA or slows its progression. Several clinical trials are under way to ease the disease’s symptoms. “Unfortunately,” Poston says, “most [trials] in the past have failed.”
But when Looney did his own research, he learned one person with the disease had lived 17 years past diagnosis. He wanted to see his two sons, ages 12 and 15 in 2007, graduate from high school. So he decided to learn everything he could to live longer with MSA. “I dove into this,” he recalls. Soon, he discovered: “Wow, this is an amazing world.”
Spinning mice
As Looney began his research, Prusiner’s group began learning more about MSA as well.
The Prusiner lab at UC San Francisco’s Institute for Neurodegenerative Diseases consists of long, interconnected galleries with rows of lab benches overlooking the city’s downtown. Visitors don disposable blue jackets and plastic gloves in a receiving chamber. The team shares no equipment with other labs to prevent contamination.
A postdoctoral researcher named Amanda Woerman joined the lab in 2013 to study proteins associated with Alzheimer and Parkinson. In both diseases, different types of protein were refolding and accumulating to form tangles in the brain. A few years earlier, scientists had found evidence that these prion-like tangles could spread to uninfected brain tissue transplants received by Parkinson patients.
 |
| Photo: Natalie Jacewicz |
| Researcher Amanda Woerman outside her lab at UCSF. |
|
The lab’s researchers had set out to prove that Parkinson tissue was contagious, but they quickly ran into a problem. After injecting mice with liquefied Parkinson brain tissue—what Woerman calls a “brain milkshake”— the scientists could not identify a single sick mouse. The experiment had failed.
But they found something even stranger. While the Parkinson mice seemed healthy, two other groups of mice did not. They staggered with an uncoordinated gait and spent most of their time whirling in pirouettes. When researchers scanned the brains of the mice, they discovered images alight with green blobs: masses of protein.
These two groups had received a different brain cocktail. The scientists had chosen tissue from a brain with MSA, a disease never shown to be infectious. Now, the team had stumbled on a discovery: Perhaps Parkinson wasn’t contagious, but MSA brain tissue was.
Testing an accident
Prusiner and his researchers knew their accidental discovery could have major ramifications. In the time since they had shown PrP proteins could morph into prions, scientists had linked no new prions with neurodegenerative disease. But the MSA mice showed clusters of a different protein called “alpha-synuclein.” Healthy brains contain this protein, but its function remains unknown.
Kurt Giles, one of the scientists involved in the first Parkinson study, began leading a team to replicate the results.
 |
| Photo: Natalie Jacewicz |
| UCSF researcher Kurt Giles in his office. |
|
Soft-spoken with a British accent, Giles maintains a neat office near the lab. An old map of Oxford University, his alma mater, hangs in the corner. If asked to draw a picture of a misfolded prion, he says he can’t. Scientists are still trying to figure out what the individual structures of prions look like. Just as the same sheet of paper can crumple into multiple shapes, prions from the same type of protein might twist into different forms and cause different diseases.
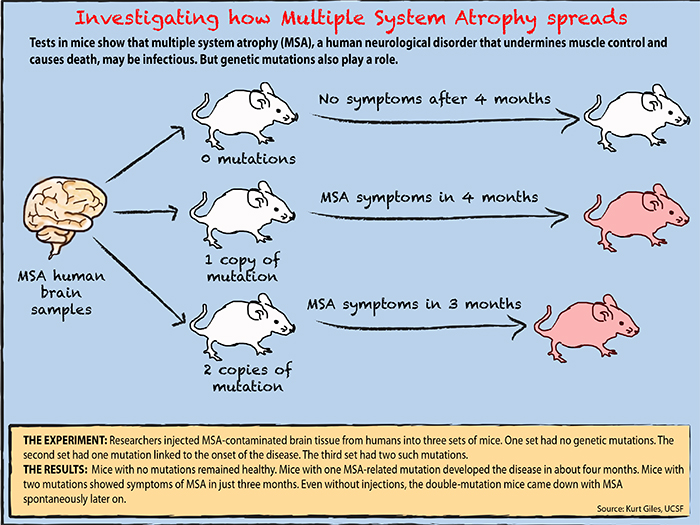
In the first experiment, the scientists had used mice with a tiny genetic mutation sometimes found in Parkinson patients. By using mice predisposed to Parkinson, the researchers had hoped they’d be more likely to succeed in transmitting the disease from infectious tissue. Instead, they apparently uncovered a genetic vulnerability to MSA.
In the replication experiment led by Giles, 12 groups of mice with the same single mutation received MSA brain tissue. But the team also put MSA tissue in mice with two copies of the mutation, as well as in some mice with no mutation at all.
The mice with no mutations developed no symptoms. But after four months, mice with one copy of the gene mutation began showing signs of neurodegeneration. Masses of alpha-synuclein prions cluttered their brains. The mice with two copies of the genetic mutation showed symptoms in even less time: three months.
The team had confirmed the initial experiment’s results. A genetic error seemed to predispose alpha-synuclein proteins to misfolding, and other prions could trigger the process. Giles and his team had discovered a new prion.
The finding excited scientists who suspect that prions cause several diseases. “I’m not surprised at all,” says Glenn Millhauser, a biochemist researching prions at UC Santa Cruz. “It speaks to Prusiner’s vision.”
 |
| Graphic: Natalie Jacewicz |
| Click on image to view larger version. |
Growing a disease
Still, the experiment wasn’t perfect. “Everyone accepts that these models are far from ideal,” Giles says of the mice.
For one thing, MSA is a disease that occurs naturally in humans, not mice. Scientists can re-create the symptoms in mice, but they can't study how mice develop the disease without human intervention. What’s more, many neurodegenerative diseases take years, even decades, to unfurl in humans—well beyond a mouse’s life span. Mice might die naturally before symptoms even appear. Finally, mice brains lack entire regions found in human brains.
That’s where Amanda Woerman came in. To study the prions effectively, scientists needed to isolate them from the muck of compounds that swamp brain tissue and test them in human cells. Woerman worked on transmitting isolated prions to human embryonic kidney cells, which grow faster than neurons. Prusiner’s lab had perfected a technique for isolating a different type of protein and injecting it into cell samples. Using a giant automated microscope, Woerman had run tests on those samples to spot clumping proteins chemically treated to glow green.
Now Woerman applied the same technique to alpha-synuclein prions in fast-growing human cells. She transmitted the prions to 18 samples from an immortalized line of human kidney cells. After four days, all of them were infected by prions. Glowing blobs lit up the cells where the misfolded proteins had spread.
Linking prions to MSA bolstered Prusiner’s claims about their role in Parkinson and Alzheimer as well. Though Parkinson doesn’t exhibit clusters of PrP, it does involve clusters of alpha-synuclein. If these proteins operate as prions in MSA, they might do the same in Parkinson.
Woerman’s work also challenged prion critics who argue that infectious viruses, rather than prions, cause these diseases. Woerman had treated the proteins with chemicals that break down DNA and RNA before transmitting them to cells, so the protein blobs that formed could not have arisen from a DNA-bearing virus.
“In science, nothing ever works the first time,” Woerman says. “But the synuclein assay was just beautiful.”
The persistence of memory
Still, some scientists doubt the claims. Jeffrey Kordower, a neurosurgeon who studies prions at Rush University in Chicago, estimates that only one-quarter of doctors and neuroscientists believe prions underlie most neurodegenerative illnesses. Just using the term “prion-like” can raise eyebrows among colleagues when submitting papers for publication, he notes.
Critics raise several issues with attributing MSA to prions. For example, researchers thus far have failed to infect normal, nonmutant mice with MSA. Not all humans who contract MSA have gene mutations, so theoretically, if they truly trigger disease, prions also should infect nonmutant mice. One recent experiment at the University of Florida suggested that prions might not cause protein clusters at all. Scientists injected mice with healthy prion-free brain tissue. The mice developed masses of protein all the same.
But even scientists with reservations about the MSA study admit they find the work interesting. The conversation has progressed since Prusiner first proposed that prions exist, says Millhauser of UC Santa Cruz. Just a few researchers still believe neurodegenerative diseases are caused by viruses, even if they have other reservations, Millhauser comments: “There are people out there who will push back on anything Prusiner says.”
Giles argues many scientists agree with the Prusiner lab’s fundamental claims that these proteins exist and self-replicate. But they may have narrower definitions of “prion,” such as applying it only to proteins contagious in day-to-day circumstances. “It’s a knee-jerk reaction to the term,” Giles says of prion critics.
Some researchers simply resist ascribing disease to prions more broadly. Giles points out that people have studied prions in yeast for years, and “no one’s ever complained.” Other researchers, with little controversy, have claimed prion-like refolding may have an upside: helping the brain retain memories. But Giles says whenever scientists blame prions for disease, “everyone throws their hands up.”
According to Giles, prions could give researchers and doctors more tools to treat patients, rather than constraining them. “What this is really leading to,” he says, “is a new way of looking at these diseases. If new therapeutics come from this, that’s definitely the goal of basic research.”
Eight years and counting
Tom Looney saw the group’s study when it came out in 2015 in Proceedings of the National Academy of Sciences. He now serves as the first patient to sit on the Board of Directors for The MSA Coalition, a national charity funding MSA research.
 |
| Photo courtesy of Tom Looney |
| Tom Looney at his home. |
|
Looney does not seem like someone who has lived with MSA for eight years. One doctor refused to believe Looney had MSA until running advanced tests of his autonomic nervous system, which the disease degrades. Looney is characteristically sunny as he describes being X-rayed when he swallowed liquid barium: "One of the coolest tests! You can literally see your insides."
Over time, Looney says, he has learned to “manually” execute body functions that work automatically in healthy people. Thanks to speech therapy, his slur is no worse than the average lazy English speaker’s. Rather than cut back on exercise, Looney increased walking and learned to maintain his balance by sucking in his stomach when he stood up. His diet is healthier, and he no longer drinks alcohol.
In January 2016, Looney’s wife called 911 after he passed out twice while seated in their North Carolina home. Looney didn’t want to call for help, but he knew it would make his wife feel better. He saw his sons finish high school. In May, his eldest son earned his college degree; his youngest will graduate in two years.
“I think it’s going to be really cool when all of this gets figured out,” he says. “I want to stick around.” After a pause, he adds, “I’m giving it my best shot.”
© 2016 Natalie Jacewicz / UC Santa Cruz Science Communication Program
Top
Biographies

Natalie Jacewicz
B.A. (biology) Harvard College
Internship: National Public Radio
The day I turned 11, my father roused seven friends and me at 6 a.m. to come see “a natural phenomenon” in the back yard. We tripped out of our sleeping bags and raced outdoors, expecting a solar eclipse or incoming meteor. Instead, we found two dung beetles rolling a clump of golden retriever scat. We went back to sleep.
Communication drives whether we celebrate a scientific discovery or wipe it off our shoes. Soon enough, nature’s rich stories drew me in. The evolutionary tale of Anolis lizards seduced me into a biology degree. My policy interests led me to management consulting in Washington D.C., where I learned how drug companies prioritize research and how NGOs select projects.
Science and policy profoundly shape each other. I want to awaken readers to the intricacies of both.
Natalie Jacewicz’s website
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .

Leigh Nelson
B.A. (English, concentration in Gender and Women’s Studies)
B.M. (vocal performance) Oberlin College
Certificate (pre-medical studies) Mills College, Oakland
Certificate (botanical illustration) Cornell University
I graduated from Oberlin and began a master’s of music with the intent of singing opera professionally. When that didn't look so promising, I made a giant academic U-turn into a two-year pre-med program. Mills strived to devour my soul because the skills I brought to the table (writing, creativity, art) were completely useless in a pre-med environment. During my second year, to save myself from utter despair while I toiled through two semesters of physics and organic chemistry, I took an advanced drawing class.
Later, as I reflected on my experience at Mills, I realized that my favorite part was that drawing class. My second favorite class was biology lab, because I got to draw things. This did not bode well for a career in medicine, but it did point the way toward medical and natural science illustration, where my interest in science could productively co-exist with my predilection for visual presentation. I have continued to perform with professional opera companies on the side.
Leigh Nelson’s website
Top |

